Li+, Na+, K+ Size increases down a group as more shell are being added.į-, O2-, N3- Size increases as nuclear charge decreases in the same period/isoelectronic. 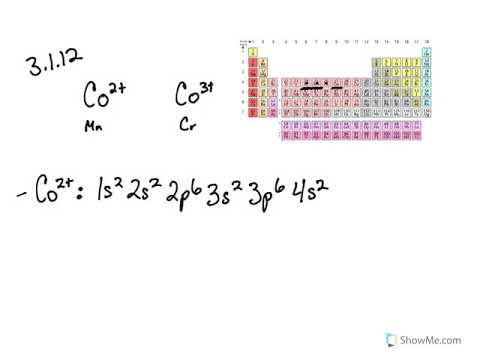
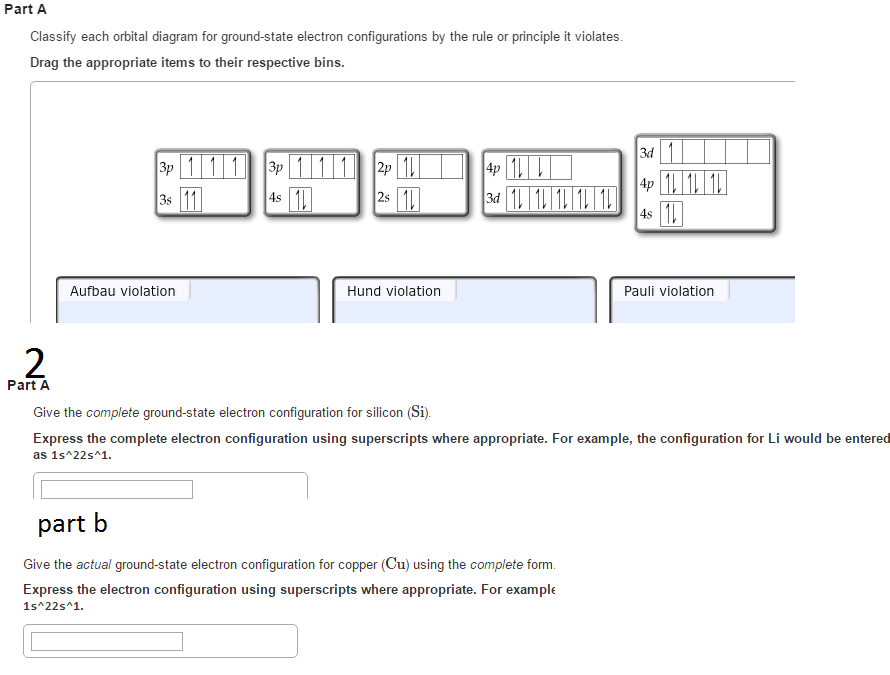
In (d), only few candidates correctly arranged the ions in order of increasing size with a reason as follows: In (c)(iii), candidates correctly gave nuclear fusion, nuclear fission and half – life respectively, for each of the nuclear processes. In (c)(ii), they were able to give the charge on each of the radiations as electrically neutral and positively charged respectively. Electronic configuration of Copper: The atomic number of Copper ( Cu) 29 Therefore, the expected electronic configuration is Ar 3 d 9 4 s 2. In (c)(i), most candidates knew that gamma rays penetrate lead block and alpha particles will be stopped by thin paper. Electronic configuration: Electronic configuration is the distribution of the electrons in orbitals of atoms using some basic principles like the Pauli exclusion principle and the Aufbau principle. In (b)(iv), candidates were able to write Cu2O/CuCl/Cu2Cl2 as the formula of one compound of Cu+. In (b)(iii), most candidates wrote either “reduction” or “oxidation” instead of redox or disproportionation In (b)(ii), majority of the candidates did not know that Cu+ had no unpaired electron and Cu2+ had only one unpaired electrons. Ground state means that the atom has the lowest energy allowed. The expected response from candidates was as follows: Electron configuration notation provides us with information about the basic energy levels and sublevels that electrons occupy. In (b)(i), only few candidates correctly wrote the electronic configuration of Cu+ and Cu2+ (ii) Electrons occupy each orbital singly first before pairing takes place in a degenerate orbital/the most stable arrangement of electrons in subshells is the one with the greatest number of parallel spins. (i) Two electrons in the same orbital of an atom cannot have same values for all four quantum numbers/no two electrons can have the same four quantum numbers/ no two electrons in the same orbital of an atom can have the same spin. In (a)(i) and (ii), candidates correctly stated Pauli’s Exclusion principle and Hund’s rule of maximum multiplicity as follows: This question was attempted by majority of the candidates and the performance was good. (e) Determine the percentage composition of phosphorus and oxygen in Give a reason for your answer in each case. (d) Arrange the following ions in order of increasing size. Time required for one-half of the atoms of a radioactive substance to decay Splitting of a heavy nucleus into two or more lighter nuclei 
Combination of two lighter nuclei to form a heavy nucleus (iii) What term is used to describe each of the following nuclear processes? (ii) Give the charge on each of the radiations mentioned in (c) (i) Name the type of radiation that will (iv) Write the formula of one compound of Cu+. (iii) State the type of reaction represented by the following equation: (ii) Give the number of unpaired electrons in each of the ions in (b) Write the electronic configuration of each of the following ions of copper: We know from the periodic table that copper is in group 11, so this agrees with the result of this electron configuration calculator.(ii) Hund’s rule of maximum multiplicity. Let us look at the electron configuration for helium, which is 1 s 2 1 4s^1 3 d 10 4 s 1. 
The electron configuration is responsible for many physical and chemical properties of an element. Electron configuration notation provides us with information about the basic energy levels and sublevels that electrons occupy.
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
